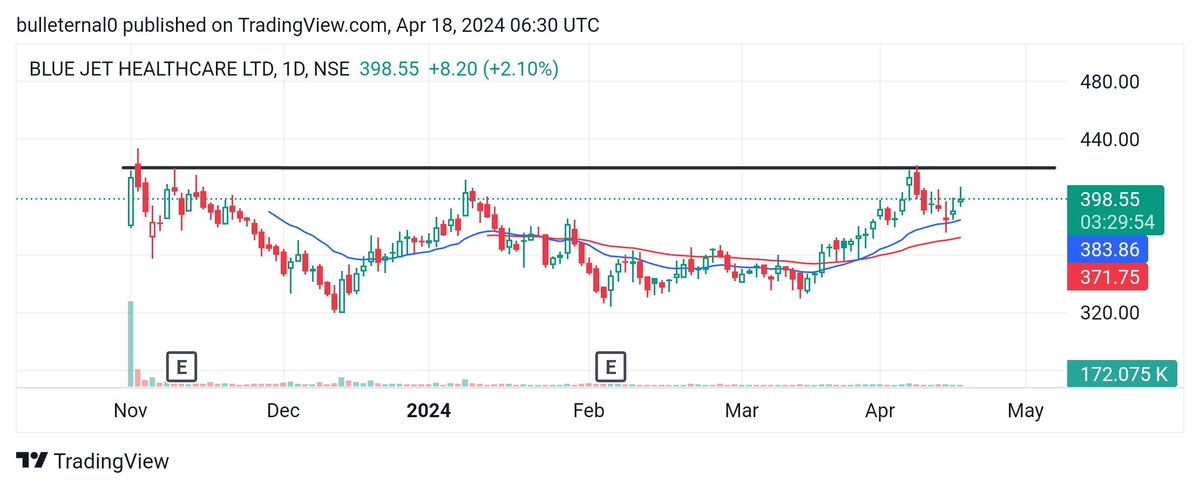
Good news for Neuland Labs.
USFDA has approved Esperion's new label expansions for NEXLETOL (bempedoic acid) & NEXLIZET (bempedoic acid & ezetimibe) for cardiovascular risk reduction
More than 70 million patients to now be eligible for NEXLETOL and NEXLIZET in US
#neulandlabs




#يحدث_الآن
⚡️Join us in exploring the article exploring the efficacy of Bempedoic acid in reducing major adverse cardiovascular events in statin- intolerant patients✨💫
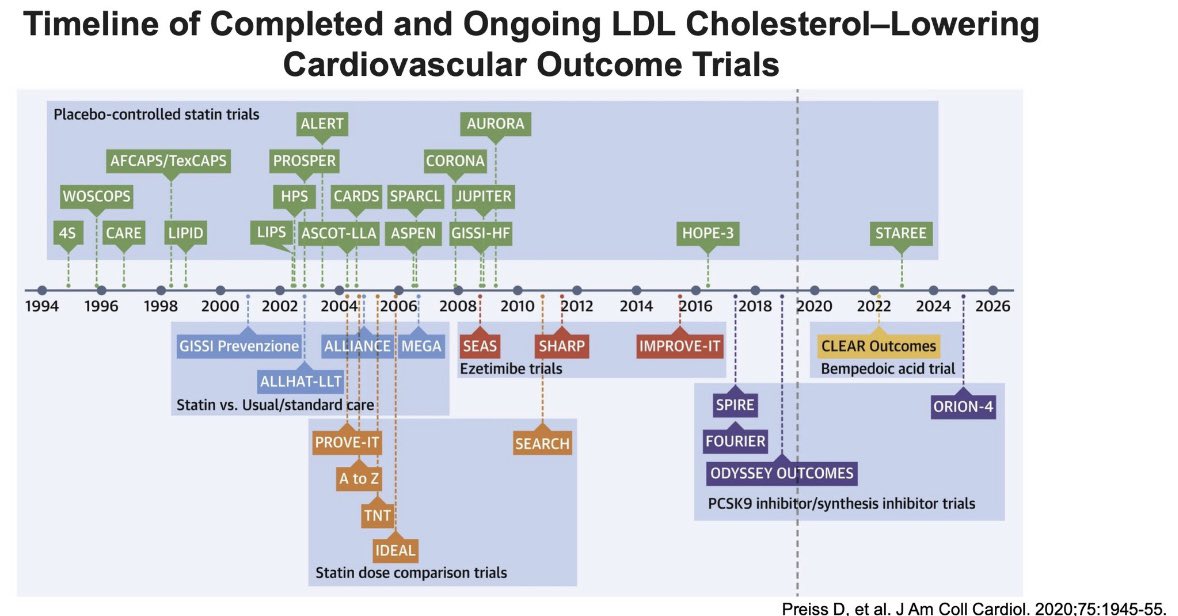
Topic : Article Alert Topic 3: Bempedoic Acid and Cardiovascular Outcomes In Statin-Intolerant Patients
By







🔴Effects of bempedoic acid on markers of inflammation and Lp(a) #2024Review #openaccess
journals.lww.com/co-cardiology/…
#CardioEd #Cardiology #MedEd #ENARM #cardiotwiteros #meded #cardiology #CardioTwitter #CardioEd #medtwitter

$ESPR New reports that 3 major payers are board June 1 to cover bempedoic acid and many moving up their drug tier status non that the label is expanded for both primary& secondary prevention. The only non statin for primary prevention
#CardioTwitter
#Statinintolernce
#Diabetes







Is your practice taking care of patients facing #statin -intolerance? #primarycare
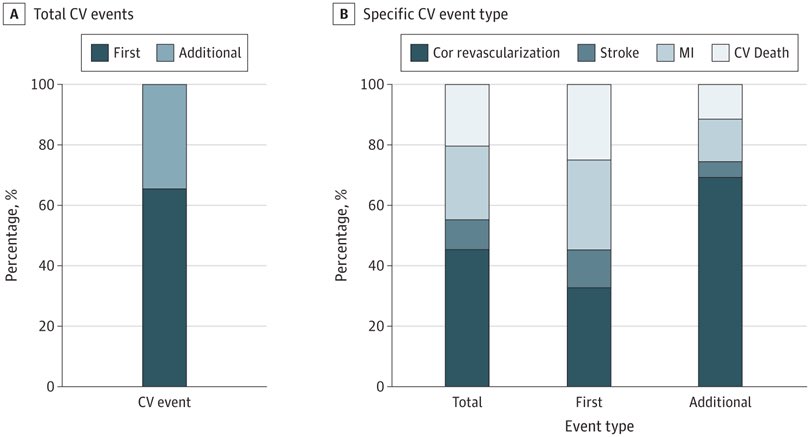
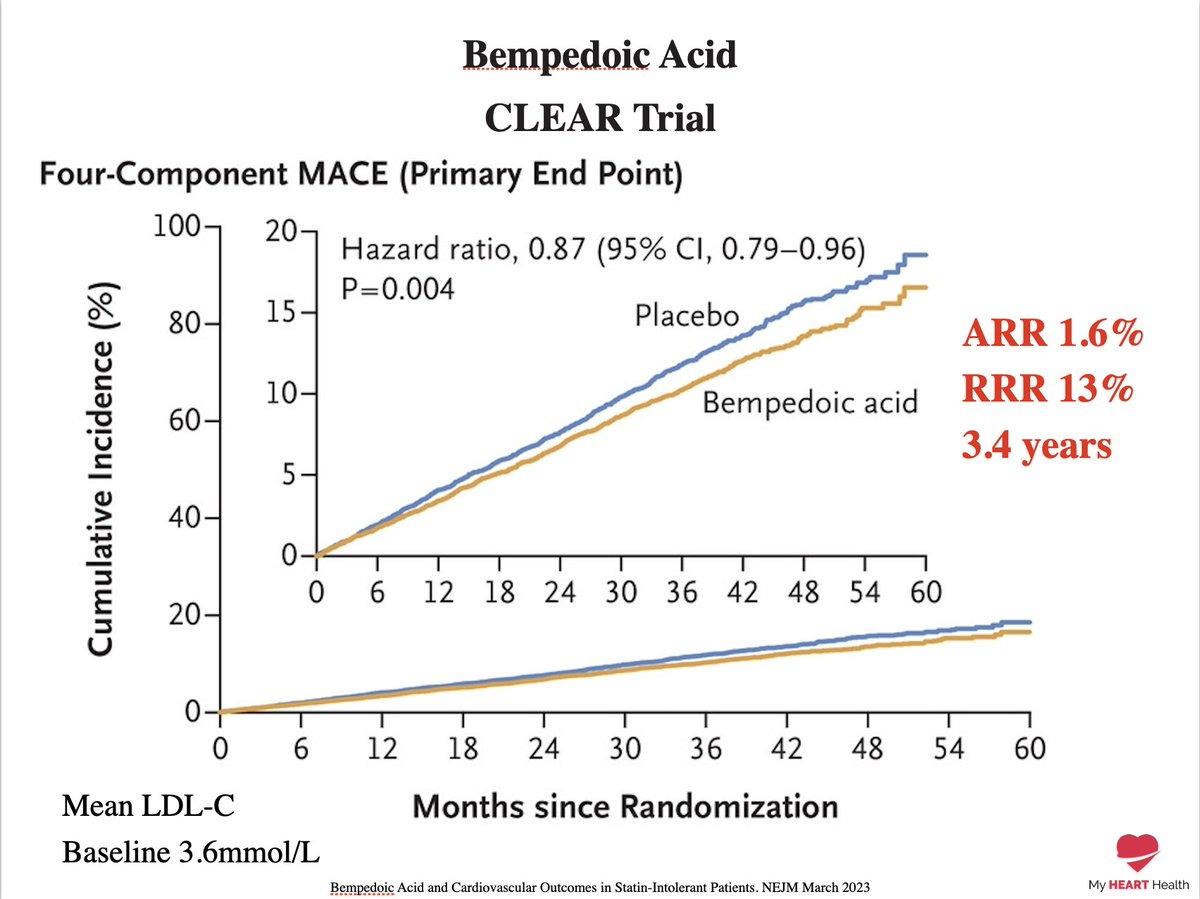
In a long-term study, bempedoic acid (Nexletol) significantly reduced the risk of heart attacks, strokes, and other cardiovascular events (39.9 months). #BempedoicAcid #HeartHealth