

Delighted that our next paper using 2+2 cycloaddition to make BCHs with 11 (!) different substitution patterns has today been accepted in Chemical Science! Congratulations to Marius and Justin and big thanks to Christopher from Alcarazo Lab for his usual excellent X-rays!


A new manuscript now on ChemRxiv - a DyKAT process discovered by Aífe with computational insight from Ali Goodfellow
St Andrews School of Chemistry
ChemRxiv
De-epimerizing DyKAT of β-Lactones Generated by Isothiourea-Catalysed Enantioselective [2+2] Cycloaddition - go.shr.lc/3vTJme2
![Andrew Smith (@ADS10_StAndrews) on Twitter photo 2024-01-29 21:22:23 A new manuscript now on ChemRxiv - a DyKAT process discovered by Aífe with computational insight from @ali_goodfellow
@StAndrewsChem
@ChemRxiv
De-epimerizing DyKAT of β-Lactones Generated by Isothiourea-Catalysed Enantioselective [2+2] Cycloaddition - go.shr.lc/3vTJme2 A new manuscript now on ChemRxiv - a DyKAT process discovered by Aífe with computational insight from @ali_goodfellow
@StAndrewsChem
@ChemRxiv
De-epimerizing DyKAT of β-Lactones Generated by Isothiourea-Catalysed Enantioselective [2+2] Cycloaddition - go.shr.lc/3vTJme2](https://pbs.twimg.com/media/GFCi9xvWwAAvjYP.jpg)




Hi LatinXChem, presenting my work ‘Ozone Conversion & CCN precursors formation from BVOCs in the troposphere by 1,3-Dipolar Cycloaddition’ at #LatinXChem23 #LatinXChemComp #Comp142
PCCP


Eveline Tiekink (Eveline Tiekink) of Vrije Universiteit Amsterdam The TheoCheM Group presents a contributed talk ESOR2023 YRS on regioselectivity in cycloaddition reactions with tropone. #ESOR2023


Hi LatinXChem community! This is my work 'An expedite and regioselective synthesis of 3,4-diaryl-1H-pyrazoles through a [3+2] cycloaddition' at #LatinXChem23 #LatinXChemOrg #Org113 OBC EurJOC.
This work was done at Instituto de Química
DOI: doi.org/10.1039/D3OB00…
![Carlos Daniel García Mejía (@CarlosDgm24) on Twitter photo 2023-10-16 12:30:38 Hi @LatinXChem community! This is my work 'An expedite and regioselective synthesis of 3,4-diaryl-1H-pyrazoles through a [3+2] cycloaddition' at #LatinXChem23 #LatinXChemOrg #Org113 @OrgBiomolChem @EurJOC.
This work was done at @iquimicaunam
DOI: doi.org/10.1039/D3OB00… Hi @LatinXChem community! This is my work 'An expedite and regioselective synthesis of 3,4-diaryl-1H-pyrazoles through a [3+2] cycloaddition' at #LatinXChem23 #LatinXChemOrg #Org113 @OrgBiomolChem @EurJOC.
This work was done at @iquimicaunam
DOI: doi.org/10.1039/D3OB00…](https://pbs.twimg.com/media/F8j6ODdXAAAJaxo.jpg)


Dr. Biplab Maji delivered an excellent talk during the Panacea Biotech Session on the final day of #NOSTOCC2024 , discussing 'Dearomative Cycloaddition Reactions via Visible Light Energy Transfer Catalysis'. #Catalysis Biplab Maji IISER Kolkata


Exciting news! Dr. Biplab Maji Biplab Maji from IISER Kolkata will discuss 'Dearomative Cycloaddition Reactions via Visible Light Energy Transfer Catalysis' at #NOSTOCC2024 . Join us for this illuminating session! #Chemistry #Catalysis #IISERKolkata



Transition metal-free, electrochemical [2+2+2]-cycloaddition: Homo(!)- and Hetero(!!)coupling of two (different(!!)) alkynes and nitriles: batch or flow. Congrats Mangish, Tirtha, and Mattia; a great collaboration with
Barham Lab and Rehbein Research. dx.doi.org/10.26434/chemr…
![Reiser-group (@reiser_group) on Twitter photo 2024-04-22 22:36:24 Transition metal-free, electrochemical [2+2+2]-cycloaddition: Homo(!)- and Hetero(!!)coupling of two (different(!!)) alkynes and nitriles: batch or flow. Congrats Mangish, Tirtha, and Mattia; a great collaboration with
@BarhamLab and @RehbeinResearch. dx.doi.org/10.26434/chemr… Transition metal-free, electrochemical [2+2+2]-cycloaddition: Homo(!)- and Hetero(!!)coupling of two (different(!!)) alkynes and nitriles: batch or flow. Congrats Mangish, Tirtha, and Mattia; a great collaboration with
@BarhamLab and @RehbeinResearch. dx.doi.org/10.26434/chemr…](https://pbs.twimg.com/media/GLzZovhXEAADJpy.jpg)
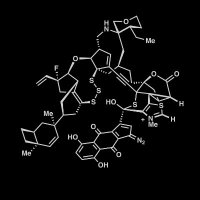
Concise #TotalSynthesis of (−)-Quinocarcin Enabled by Catalytic Enantioselective Reductive 1,3-Dipolar Cycloaddition of Secondary Amides by Kan-Lei Ji, Shu-Fan He, Dong-Dong Xu, Wen-Xin He, Jian-Feng Zheng, and Pei-Qiang Huang in Angewandte Chemie onlinelibrary.wiley.com/doi/10.1002/an…

Looking for oxazole synthesis ?
Here is an exciting catalyst-free new method using light.
Angewandte Chemie
DM Lab IIT Bombay
argha saha, Subhabrata Sen
Photoinduced [3+2] Cycloaddition of Carbenes and Nitriles: A Versatile Approach to Oxazole Synthesis
onlinelibrary.wiley.com/doi/full/10.10…
![Debabrata Maiti (@maiti_iitb) on Twitter photo 2023-11-07 12:29:49 Looking for oxazole synthesis ?
Here is an exciting catalyst-free new method using light.
@angew_chem
@dm_lab
@arghasa68168988, @SenResearchGrp
Photoinduced [3+2] Cycloaddition of Carbenes and Nitriles: A Versatile Approach to Oxazole Synthesis
onlinelibrary.wiley.com/doi/full/10.10… Looking for oxazole synthesis ?
Here is an exciting catalyst-free new method using light.
@angew_chem
@dm_lab
@arghasa68168988, @SenResearchGrp
Photoinduced [3+2] Cycloaddition of Carbenes and Nitriles: A Versatile Approach to Oxazole Synthesis
onlinelibrary.wiley.com/doi/full/10.10…](https://pbs.twimg.com/media/F-VM9AjboAAf0ZX.jpg)

Our latest paper was published in Synlett Thieme Chemistry
“Visible-Light-Induced Oxidative Generation of o-Quinone Methides for Inverse-Electron-Demand [4+2] Cycloaddition Reactions”
Congratulations Shoya and all co-authors! thieme-connect.de/products/ejour…
![Kenta Tanaka (@KENTA_TANAKA_1) on Twitter photo 2023-11-15 13:05:51 Our latest paper was published in Synlett @thiemechemistry
“Visible-Light-Induced Oxidative Generation of o-Quinone Methides for Inverse-Electron-Demand [4+2] Cycloaddition Reactions”
Congratulations Shoya and all co-authors! thieme-connect.de/products/ejour… Our latest paper was published in Synlett @thiemechemistry
“Visible-Light-Induced Oxidative Generation of o-Quinone Methides for Inverse-Electron-Demand [4+2] Cycloaddition Reactions”
Congratulations Shoya and all co-authors! thieme-connect.de/products/ejour…](https://pbs.twimg.com/media/F--iYGLbsAAd4dG.jpg)


![Chinese Chemical Letters (@ChinChemLett) on Twitter photo 2024-04-11 12:28:47 Three-step synthesis of flavanostilbenes with a 2-cyclohepten-1-one core by Cu-mediated [5 + 2] cycloaddition/decarboxylation cascade
doi.org/10.1016/j.ccle… Three-step synthesis of flavanostilbenes with a 2-cyclohepten-1-one core by Cu-mediated [5 + 2] cycloaddition/decarboxylation cascade
doi.org/10.1016/j.ccle…](https://pbs.twimg.com/media/GK4lGHgaQAEUGIO.jpg)
![Yunus Turkmen (@yunus_turkmen) on Twitter photo 2024-04-19 10:33:41 Whenever I teach the total synthesis of isocomene by Pirrung (1979) in class, I am fascinated by its efficiency and beauty. A single intramolecular [2+2] cycloaddition creates three new contiguous stereocenters all of which are quaternary!
pubs.acs.org/doi/10.1021/ja… Whenever I teach the total synthesis of isocomene by Pirrung (1979) in class, I am fascinated by its efficiency and beauty. A single intramolecular [2+2] cycloaddition creates three new contiguous stereocenters all of which are quaternary!
pubs.acs.org/doi/10.1021/ja…](https://pbs.twimg.com/media/GLhXkBtXQAAyEVQ.jpg)

![J Org Chem/Org Lett (@JOC_OL) on Twitter photo 2024-04-30 12:30:08 Learn how twisting double bonds can facilitate thermally allowed [2+2] cycloaddition reactions. Please see the latest work in #OrgLett from @JudyWuChem, @RViesser, and @MayLabUH at U of Houston. pubs.acs.org/doi/10.1021/ac… Learn how twisting double bonds can facilitate thermally allowed [2+2] cycloaddition reactions. Please see the latest work in #OrgLett from @JudyWuChem, @RViesser, and @MayLabUH at U of Houston. pubs.acs.org/doi/10.1021/ac…](https://pbs.twimg.com/media/GMabtR3WsAAgleo.png)
![Ahmad Masarwa (Masarwa Lab) (@AhmadMasarwa2) on Twitter photo 2023-11-06 10:41:14 Our latest study is now available on @ChemRxiv. In this work, we reported the regioselective synthesis of poly-borylated cyclobutanes through [2+2]-cycloaddition reactions of polyborylated alkenes. Congratulations 🥳to Nicole and @NadimEghbarieh!
chemrxiv.org/engage/chemrxi… Our latest study is now available on @ChemRxiv. In this work, we reported the regioselective synthesis of poly-borylated cyclobutanes through [2+2]-cycloaddition reactions of polyborylated alkenes. Congratulations 🥳to Nicole and @NadimEghbarieh!
chemrxiv.org/engage/chemrxi…](https://pbs.twimg.com/media/F-PmF9uWsAAVtDd.jpg)
![Organic Chemistry Portal (@organic_portal) on Twitter photo 2024-04-28 01:00:01 organic-chemistry.org/abstracts/lit7…
N,O-Acetals undergo a catalytic enantioselective [2 + 2] cycloaddition to alkenes organic-chemistry.org/abstracts/lit7…
N,O-Acetals undergo a catalytic enantioselective [2 + 2] cycloaddition to alkenes](https://pbs.twimg.com/media/GMKsv5hXIAAwbpt.jpg)