Jozefina Casuscelli
@jcasusce
MD, Department of Urology, Ludwig-Maximilians-University
ID:855801726401732609
22-04-2017 15:13:26
142 Tweets
154 Followers
124 Following

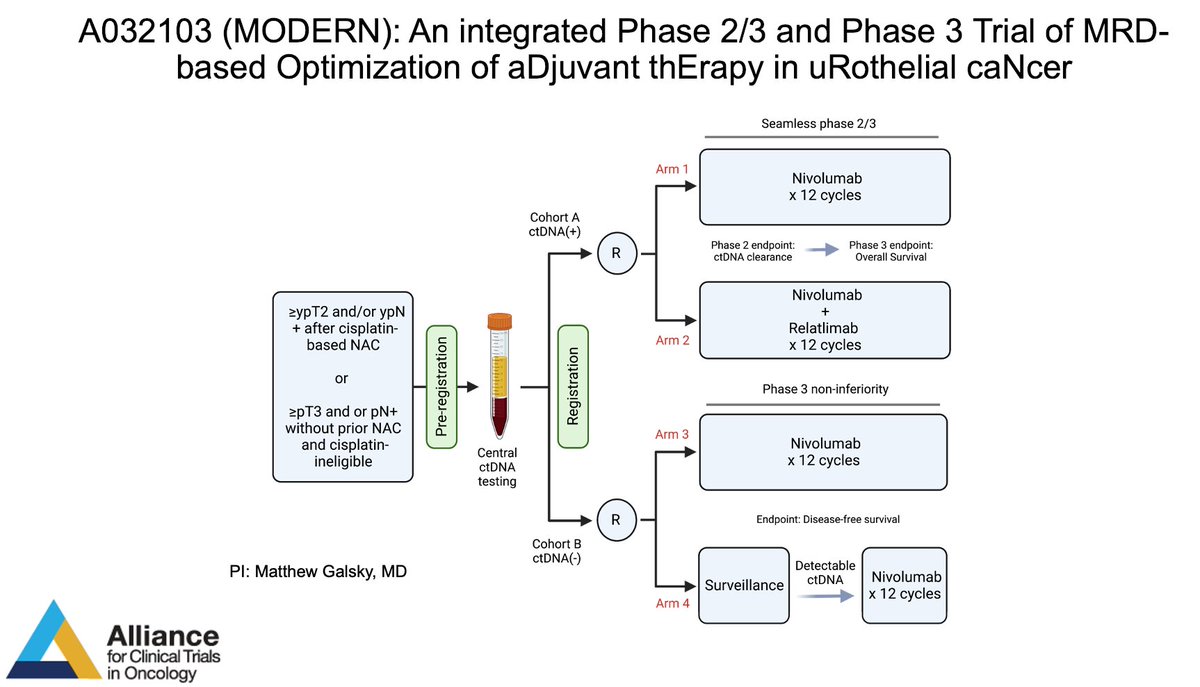
MODERN is open! Available at your site through the CRSU. Please help advance MRD-based adjuvant decision making in bladder cancer. Alliance for Clinical Trials in Oncology Alan Tan Ashish M. Kamat, MD, MBBS ECOG-ACRIN Cancer Research Group SWOG Cancer Research Network Guru P. Sonpavde, MD PaulCrispenMD Stephanie Berg Suzanne Cole, MD Nitin Yerram M.D. DrTylerStewart Jonathan Rosenberg MD

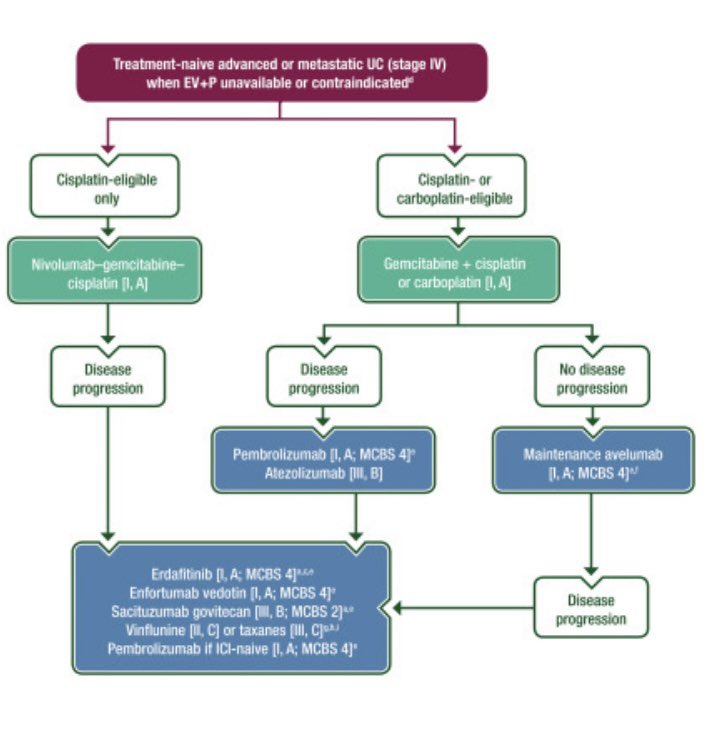
Oncology Brothers Karine Tawagi MD Sia Daneshmand, M.D. CancerNetwork® Bijoy Telivala Yakup Ergün Andrea Apolo, M.D. Santhosh Ambika Amanda Nizam, MD Petros Grivas Shilpa Gupta Enrique Grande Paulo G Bergerot The ESMO guidlines recommends EVP as the preferred standard of care, while nivo/cis and maintenance avelumab are interchangeable and recommended when EVP is not available or contraindicated

JUST IN: U.S. FDA alert: FDA approves nivolumab in combination with cisplatin and gemcitabine for unresectable or metastatic urothelial carcinoma! Via ASCO U.S. FDA alert !
Trial presented ESMO - Eur. Oncology #ESMO23 and published NEJM
OncoAlert Matt Galsky Guru P. Sonpavde, MD Michiel van der Heijden




NECTIN4 amplification predicts response to Enfortumab vedotin in mUC! It occurs frequently across solid tumors (app. 25% of mUC)➡️potential tumor-agnostic biomarker for NECTIN4-targeting therapies! Happy to share our poster K17 ASCO #GU24 ! Markus Eckstein OncoAlert UroToday.com


#Tweetorial :
Data from Drake Lab and others shows CSPC is immunologically different and not an ‘immune desert’ like CRPC. We interrogated CSPC metastases with single-cell RNA-seq (scRNA-seq), VIPER, and IF before and after tx w/ ADT +/- aPD-1 in a Phase 2 study. /1

JUST IN and again via ASCO #FDAAlerts
The FDA Oncology approved Enfortumab Vedotin ( EV) in combo with pembrolizumab for metastatic urothelial cancer!!
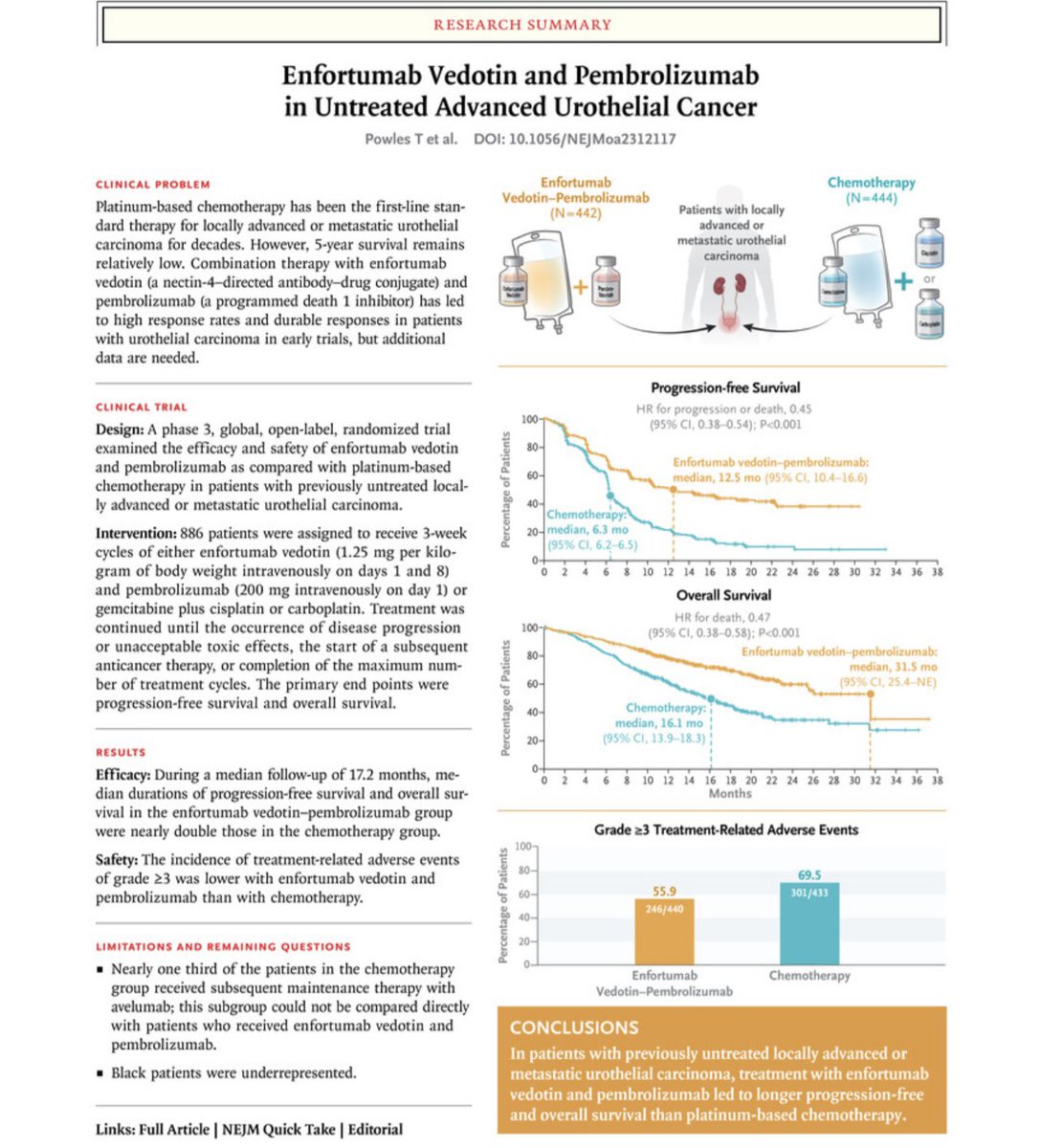
Data based on Tom Powles #ESMO23 Plenary (EV-302) #StandingOvation ESMO - Eur. Oncology
Great day for Bladder Cancer and our…



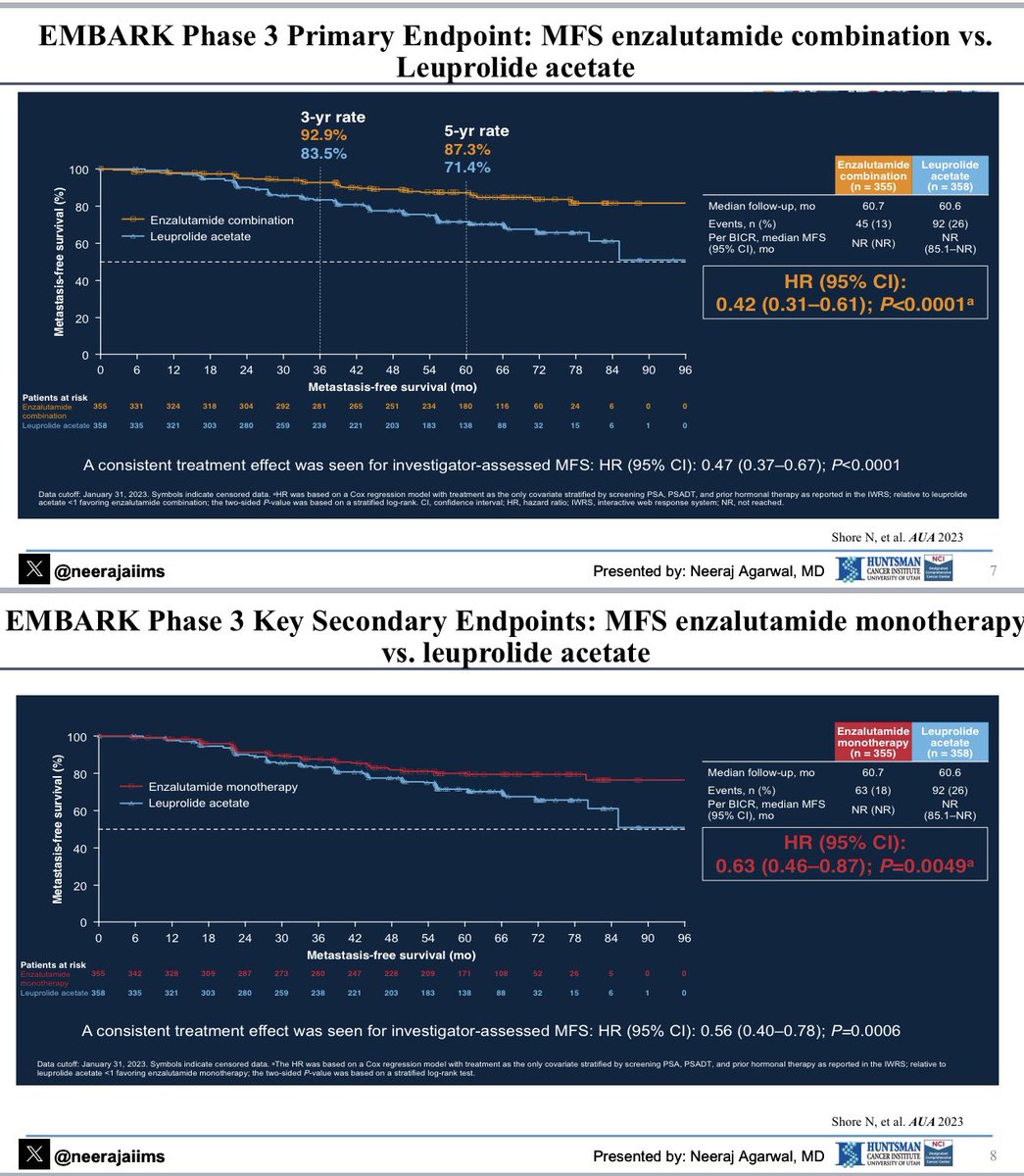
Breaking news👉 U.S. FDA approves enzalutamide with/without androgen deprivation therapy (ADT) for non-metastatic castration sensitive #ProstateCancer based on Embark trial data👇Congrats neal shore Stephen Freedland, MD Link👉 t.ly/R42C6 Prostate Cancer Foundation OncoAlert UroToday.com


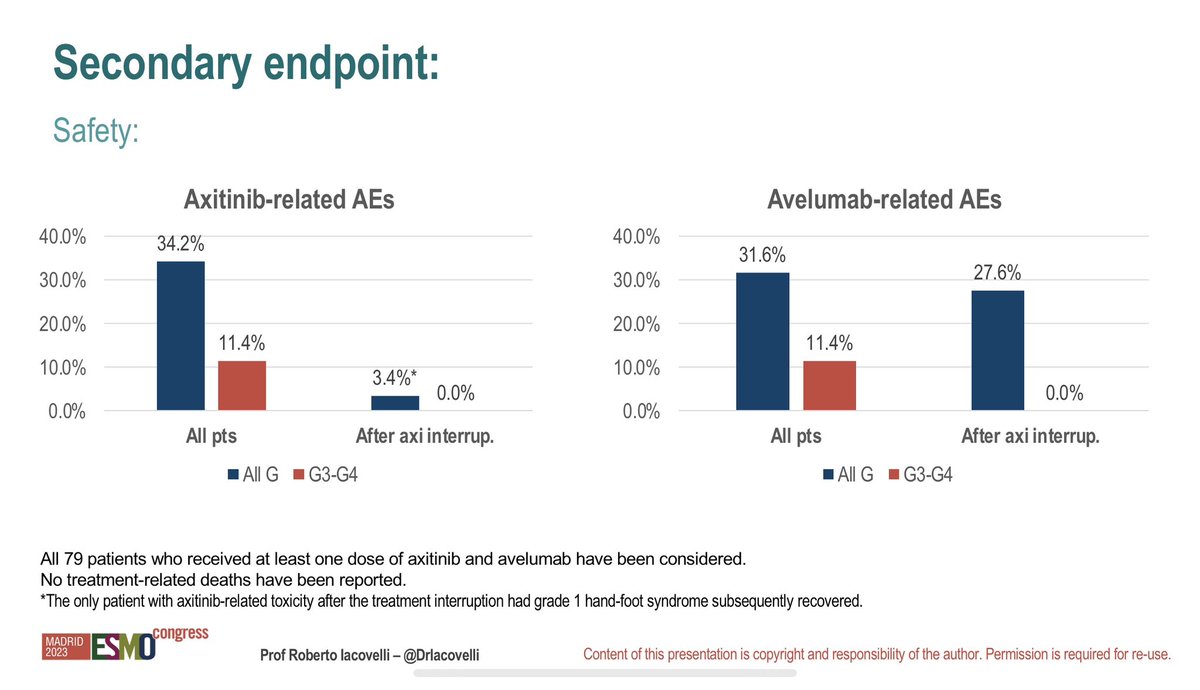
Rechallange with pembrolizumab in UC in those patients who complete 2 yrs of therapy or stop due to CR is associated with responses. A highly selected population. What does this mean in the adjuvant therapy setting? #nashvillelive Vadim Koshkin MD #ESMO2023



The ESMO - Eur. Oncology LBAs are out
And a wow for PEMBRO/EV (Presidential)
OS 31.5 vs 16.1 months (HR 0.45)
PFS 12.5 vs 6.3 months (HR 0.47)
P<0.00001
A historic change for 1L mUC since Von Der Maase JCO 2000
Tom Powles #ESMO23 OncoAlert




Adjuvant pembrolizumab significance delays disease free survival in bladder cancer. The benefit is ‘clinically meaningful’ Andrea Apolo, M.D. .It joins nivo, but not atezo, in having DFS. It’s not clear whether OS was tested-which is co-primary. Exciting times! merck.com/news/mercks-ke…

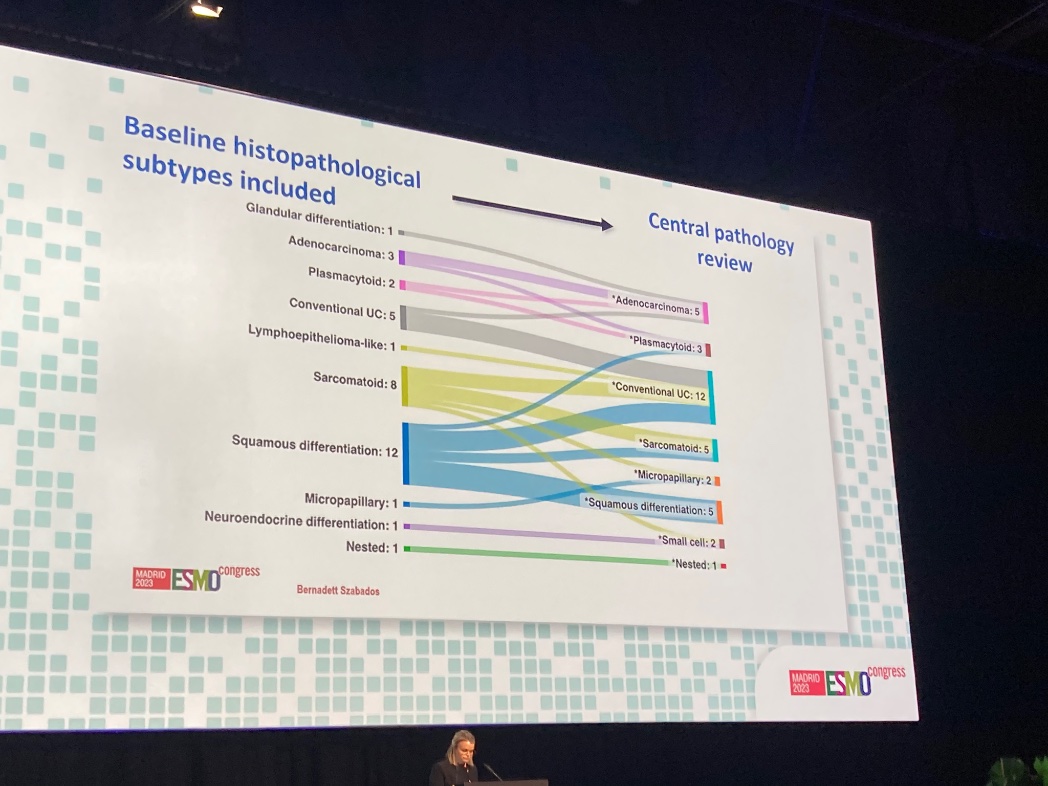
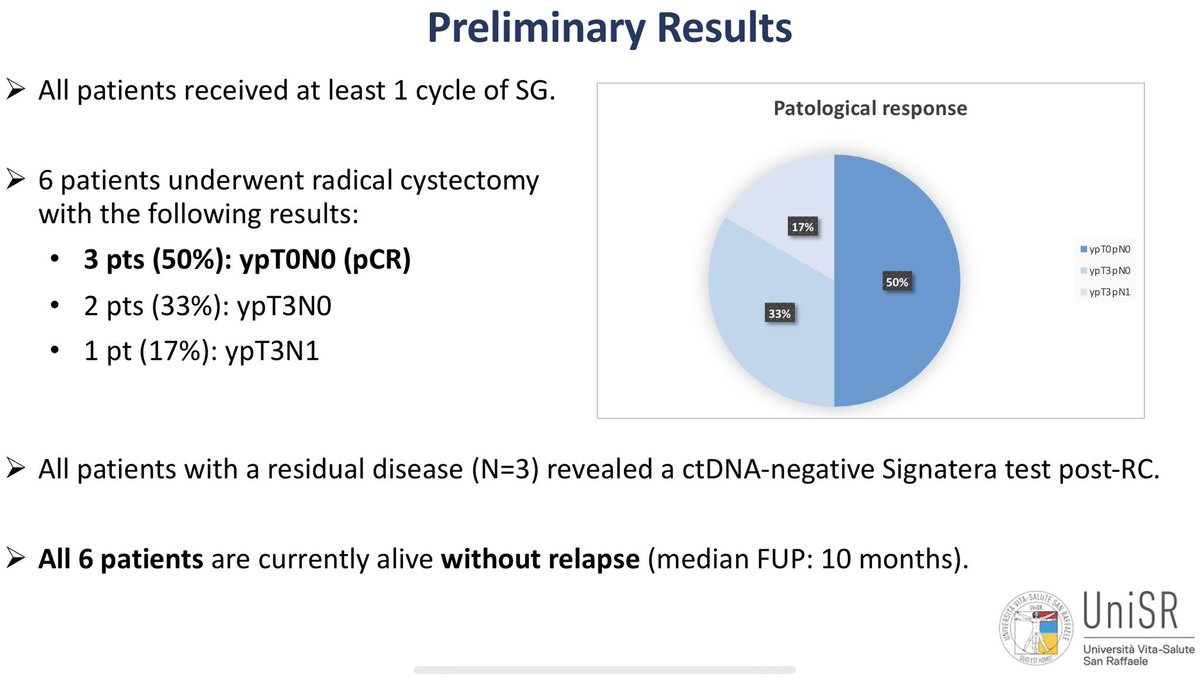
Neoadjuvant sasituzimab govitecan in muscle invasive bladder cancer (n=14) Antonio Cigliola Andrea Necchi @IBCN23 Results = a mixture of efficacy (3/6 pCRs) and toxicity (TRAE G5= GCSF/dose changes). Still early data. EV (n=22, 34% pCR, also with neoadjuvant tox). R3s with EVP

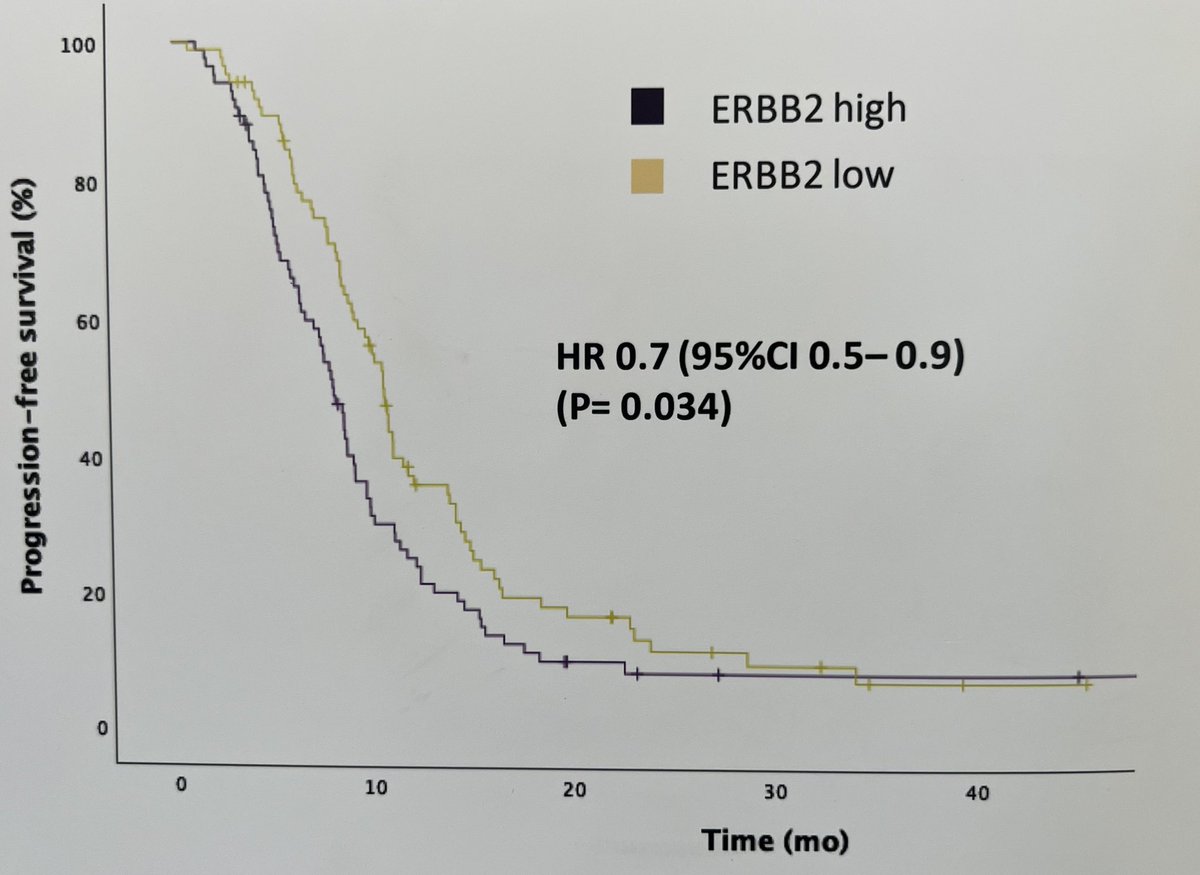
interest in HER 2 & 3 expression in urothelial cancer due to new ADCs. Exploratory analysis from R3 LAMB trial shows ~50% of tumors are HER2 and HER3 +ve. HER2 maybe associated with short PFS. Disitimab vedotin in R3 in 1st line UC, HER3 ADCs awaited in UC Bernadett Szabados @IBCN23