Rene Koenigs
@ReneKoenigs
ID:854736391418388483
http://www.koenigslab.rwth-aachen.de 19-04-2017 16:40:10
138 Tweets
1,2K Followers
658 Following

Research Highlight now online:
Forming bicycles with nitrenes by Thomas West
nature.com/articles/s4416… ($)
Highlighting the Science Magazine article by Rene Koenigs Fang Li 李方 Wenxin Felix Zhu ClaireEmpel Mykhailiuk Chem 🇺🇦 Proschak Lab and co-workers



A super-fun collaboration between Prof. Rene Koenigs and Enamine scientists O. Datsenko & Mykhailiuk Chem 🇺🇦 This study offers a surprisingly selective alternative pathway in which triplet nitrenes react with compounds bearing two double bonds! (science.org/doi/10.1126/sc…)
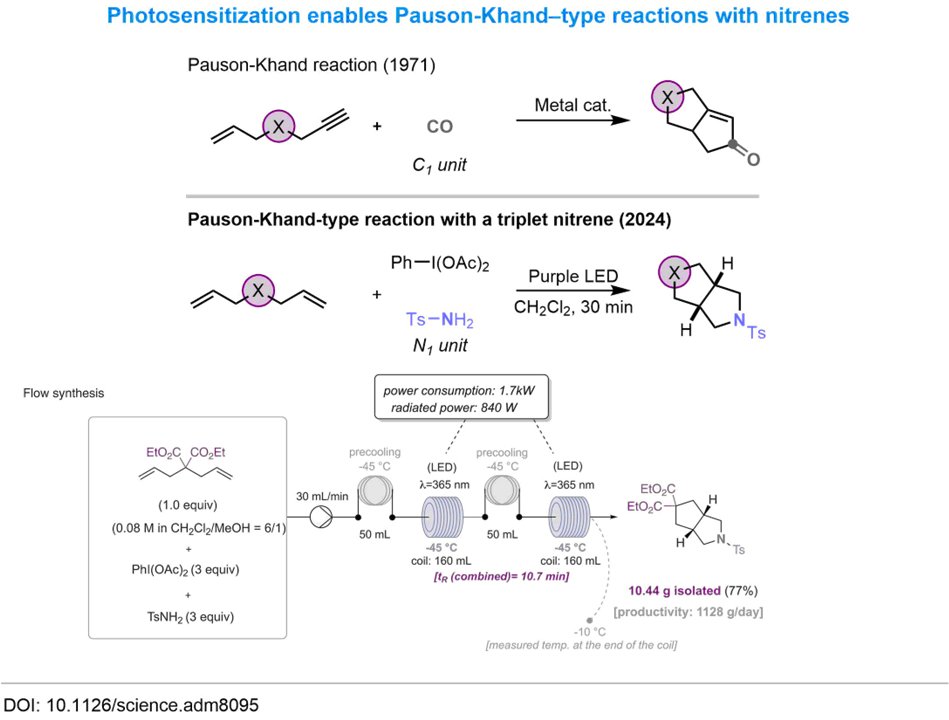

ChemRxiv ClaireEmpel Rene Koenigs Mykhailiuk Chem 🇺🇦 Proschak Lab Fang Li 李方 So trilled that the final version of this study is now published in Science Magazine!
It is about one reactivity to forge all rings: bicyclic bioisosteres of piperazine, morpholine, and piperidine. In case you are interested: science.org/doi/10.1126/sc…

SAVE THE DATE! The 1st edition of the International School in Photocatalysis and Biocatalysis ( #PHOTOCAT24 ) will be held in Padova on June 2nd-7th 2024. Fantastic line-up of speakers and amazing venue! Check it out! photocat24.com


Angew. Chem. online:
#Visible ‐Light‐Induced, Single‐ #Metal ‐ #Catalyzed , Directed C−H #Functionalization : Metal‐Substrate‐Bound Complexes as Light‐ #Harvesting Agents
Top Science @CHNSci Chemistry EurJOC
Angewandte Chemie Rene Koenigs ClaireEmpel
RWTH Aachen
onlinelibrary.wiley.com/doi/10.1002/an…

Visible-Light-Induced, Single-Metal-Catalyzed, Directed C-H Functionalization: Metal-Substrate-Bound Complexes as Light-Harvesting Agents (Koenigs) Rene Koenigs ClaireEmpel RWTH Aachen #OpenAccess thanks to #ProjektDEAL #AngewandteReviews onlinelibrary.wiley.com/doi/10.1002/an…


Rene Koenigs group demonstrates that #photocatalysis leverages amination reactions via free nitrene intermediates to allow for intermolecular amination reaction of allenes #RealTimeChem
cell.com/chem-catalysis…

We also highlight a Nature Communications paper by Rene Koenigs and colleagues that combines theory and experimentation to study photochemical and photocatalytic nitrene reactions with olefins (rdcu.be/cHl3a). nature.com/articles/s4358…

Rhodium-catalyzed CH methylation reactions with #diazoalkanes in Chemistry . TMS-diazomethane proves as a simple and readily available methylating agent. Congratulations to ClaireEmpel Sripati Jana, PhD and Tim …mistry-europe.onlinelibrary.wiley.com/doi/abs/10.100…

Divergent amination reactions with monovalent nitrogen species using visible light. Really excited to share our latest research in Nature Communications. Congratulations to Yujing and Chao! #photochemistry #RWTH nature.com/articles/s4146…

Celebrating our 1st paper of 2022, just-accepted in Angewandte Chemie, on HFIP-assisted carbonyl-olefin metathesis doi.org/10.1002/anie.2… with first author @TuongToUNSW and longtime collaborator Rene Koenigs. 2 bottles of Scotch were harmed in the process #ozchem


🌟 #SYNTHESIS highlight article🌟
Thanks to Claire Empel (ClaireEmpel), Sripati Jana (@jana_sripati) & Rene Koenigs (@ReneKoenigs).👏This review summarizes progress made on [1,2]-sigmatropic rearrangements using #carbenes in the ylide formation step👉fal.cn/3kQLv
![Thieme Chemistry (@thiemechemistry) on Twitter photo 2021-12-22 14:00:22 🌟#SYNTHESIS highlight article🌟 Thanks to Claire Empel (@EmpelClaire), Sripati Jana (@jana_sripati) & Rene Koenigs (@ReneKoenigs).👏This review summarizes progress made on [1,2]-sigmatropic rearrangements using #carbenes in the ylide formation step👉fal.cn/3kQLv 🌟#SYNTHESIS highlight article🌟 Thanks to Claire Empel (@EmpelClaire), Sripati Jana (@jana_sripati) & Rene Koenigs (@ReneKoenigs).👏This review summarizes progress made on [1,2]-sigmatropic rearrangements using #carbenes in the ylide formation step👉fal.cn/3kQLv](https://pbs.twimg.com/media/FHN5F5lXMAs7N4r.jpg)

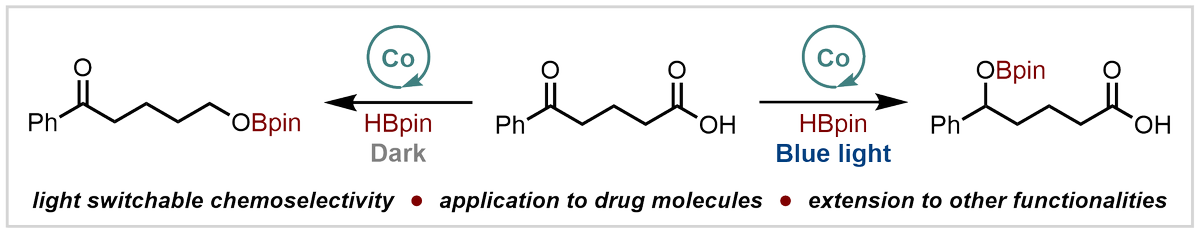
Huge congratulations to the team - our latest paper, which shows how light can control the chemoselectivity of catalysed hydroboration, is now online Angewandte Chemie
doi.org/10.1002/anie.2…

Really excited to share the latest research from our lab - C-C #coupling followed by defluorination with #carbene using #photocatalysis . Published in Angewandte Chemie thank you to Fang Li 李方 and chao pei for pushing this chemistry! RWTH Aachen #diazo

As an exclusive partner of the GDCh Science Forum Chemistry, Chemistry Europe and ChemPhotoChem are proud to announce 'Reagenzfreie Aktivierung in der organischen Synthese' with presentations by Mathias Senge, DidierResearchGroup, Rene Koenigs and many more.


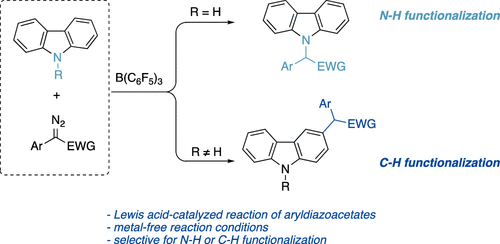
Borane-Catalyzed Carbazolation Reactions of Aryldiazoacetates (J Org Chem/Org Lett): pubs.acs.org/doi/abs/10.102….