في أول لقاح من نوعه في التاريخ ضد بعض أنواع السرطانات التي يسببها فايروس EBV. هيئة الغذاء والدواء الأمريكية FDA Oncology
تعطي الموافقة المبدئية لبدء الأبحاث السريرية في الولايات المتحدة على البشر. اللقاح يستخدم تقنية mRNA وهي نفس تقنية لقاحات موديرنا وفايزر ضد الكوفيد١٩.
جميع


Celebrating the 2024 AACR Cancer Disparities Progress Report in the US Congressional Briefing Congress with Margaret Foti, PhD, MD (hc) NCI Disparities FDA Oncology Dr. Marge Foti, Dr. Rob Winn, Dr. Sanya Springfield and Michelle Torres, daughter of Anibal Torres #hcc survivor PanOncology Trials

See how you can be a part of FDA Oncology's National Black Family Cancer Awareness Week happening next month!
#BlackFamCan

🚨HUGE victory for SCLC!🚨
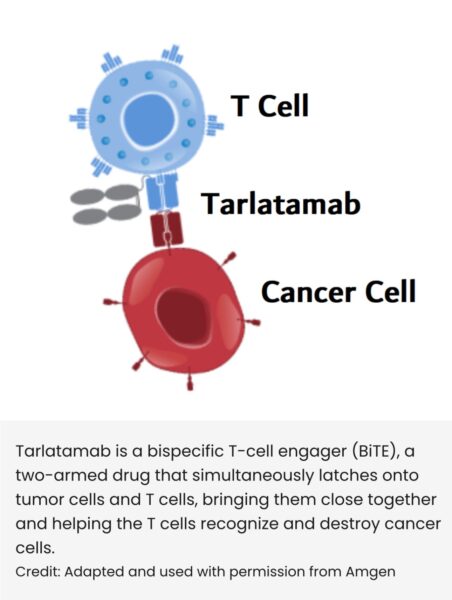
FDA Oncology, under Project Orbis and the FDA Center of Excellence, grants accelerated APPROVAL for tarlatamab (Imdelltra, DLL3 BiTE) Amgen Oncology for relapsed small cell lung cancer after platinum-based therapy!
#LCSM SmallCellSMASHERS The Shields Lab
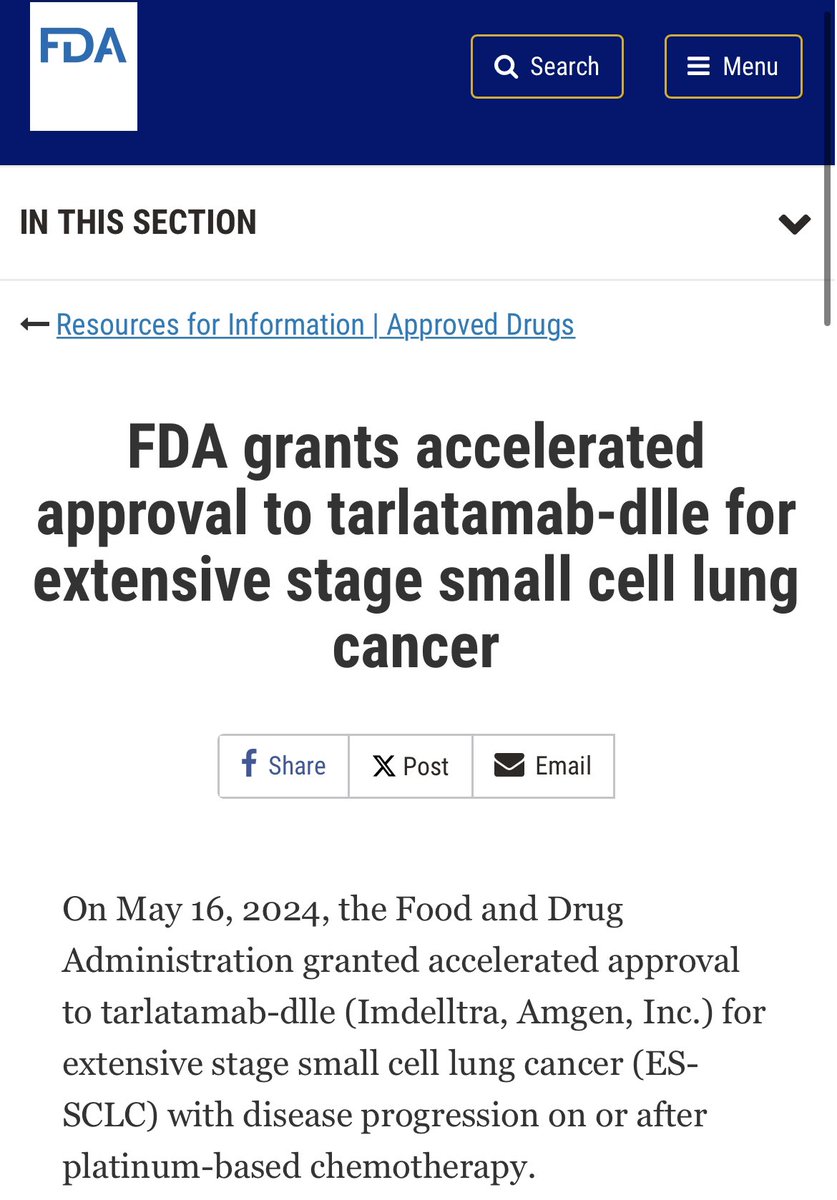


US FDA granted accelerated approval to tarlatamab-dlle - Vivek Subbiah, MD
FDA Oncology
oncodaily.com/67317.html
#Cancer #SCLC #FDAApproval #Imdelltra #OncoDaily #Oncology #SCLC #Tarlatamab

FDA Oncology Center of Excellence welcomed fellows of the FDA-AACR 2023-2024 Oncology Educational Fellowship to White Oak for a great mock ODAC and discussion!
#OCEProjectSocrates Rick Pazdur Jennifer Gao


Thanks FDA Oncology AACR for a fantastic fellowship, culminating in Project ODAC Odyssey, a mock ODAC focused on analyses of molecularly-defined subgroups in oncology clinical trials
Was fun to represent the sponsor & pleased to report ODAC voted 9-0 in our favour!
Lindor Qunaj


Please expedite #AVA6000 for metastatic breast cancer pts. “Avacta commenced a two-weekly dosing safety study in the US on the basis that this is likely to lead to better efficacy.” #AVCT #AACR24 #MBC NIH National Cancer Institute Dr. Kimryn Rathmell FDA Oncology President Biden Alexandria Ocasio-Cortez Vice President Kamala Harris avacta.com/update-on-ava6…

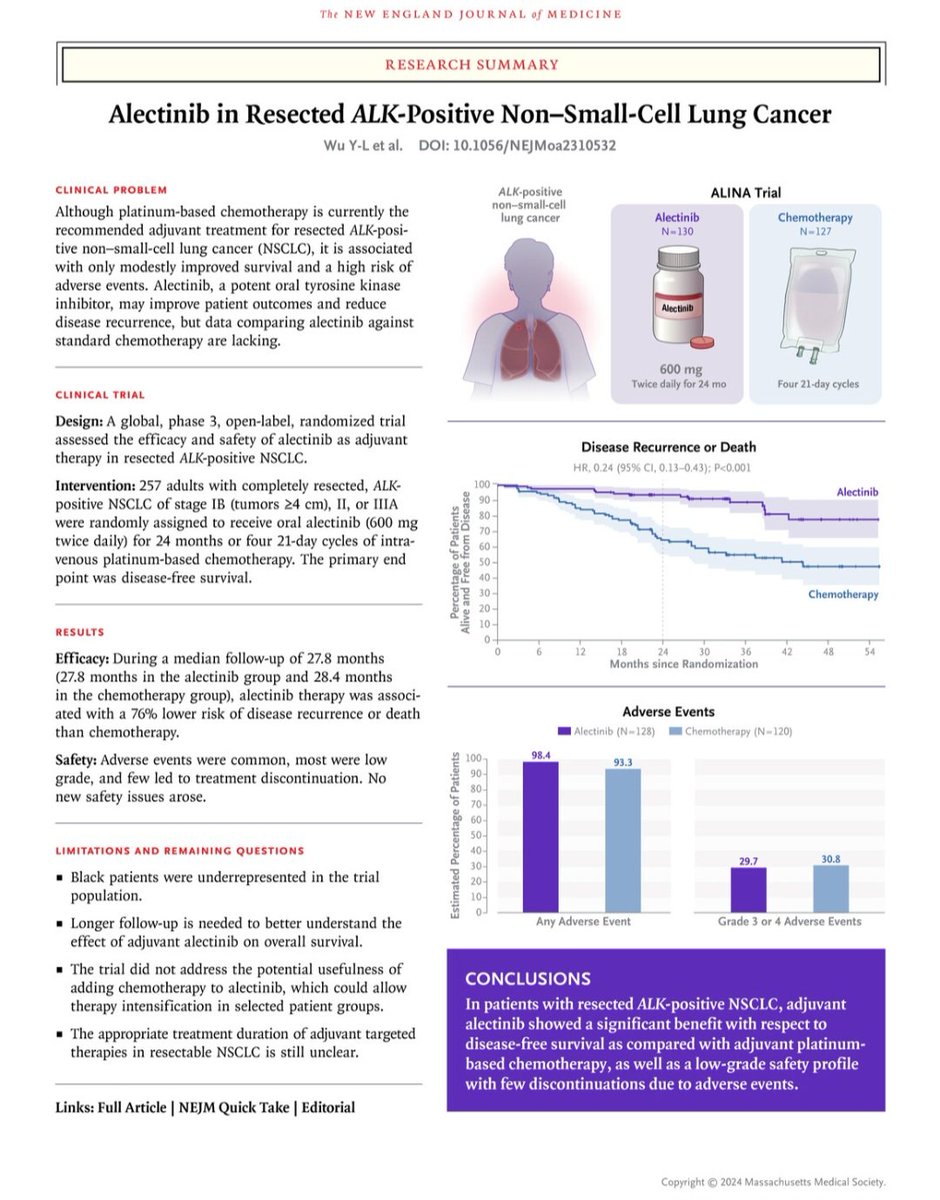
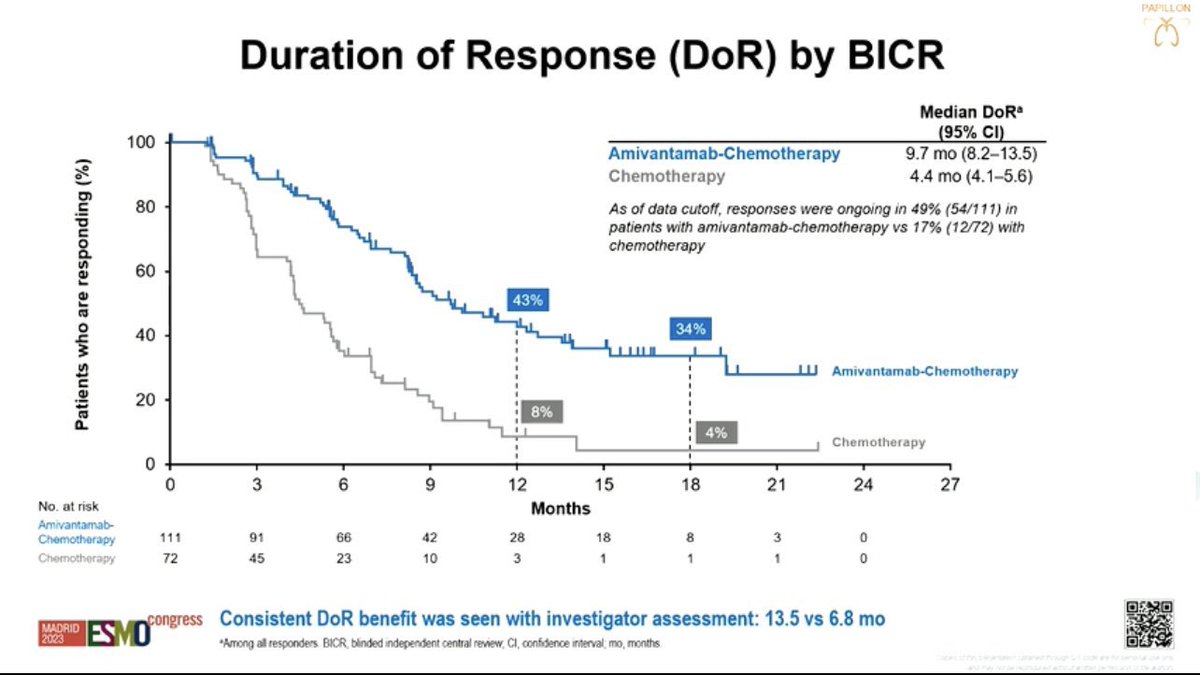
#Amivantamab now FDA Oncology approved for mNSCLC Exon20 based off #PAPPILON in 1L w/ chemo:
- PFS 11.4mos w/Ami vs 6.7mos chemo (HR: 0.40)
- ORR in 73% vs 47%
- OS favoring Ami (HR: 0.67)
- 7%⛔Ami because of AEs
- New SoC/Practice Changing
#lcsm #onctwitter #medtwitter


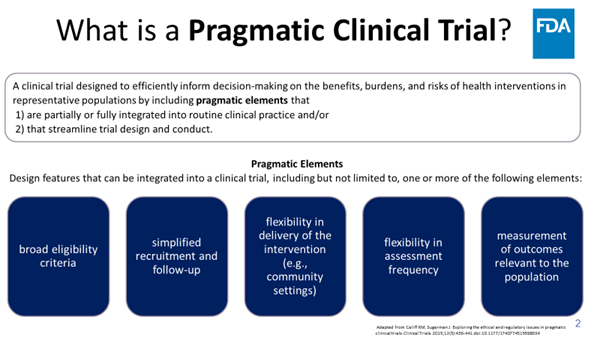
What are pragmatic clinical trials? Clinical trials that streamline data collection and design by collecting only necessary information to answer the research question. A thread 🧵 1/5
#ProjectPragmatica #OCEProject5in5 #PragmaticTrials

Thank you FDA Oncology for sharing important information in various languages (including my mother tongue Gujarati). We appreciate the efforts done to educate the community.
CMO Gujarat Narendra Modi Gujarat Samachar IndianCancerSociety
Bipin Savani Bhagirath Dholaria, MD Pankit Vachhani

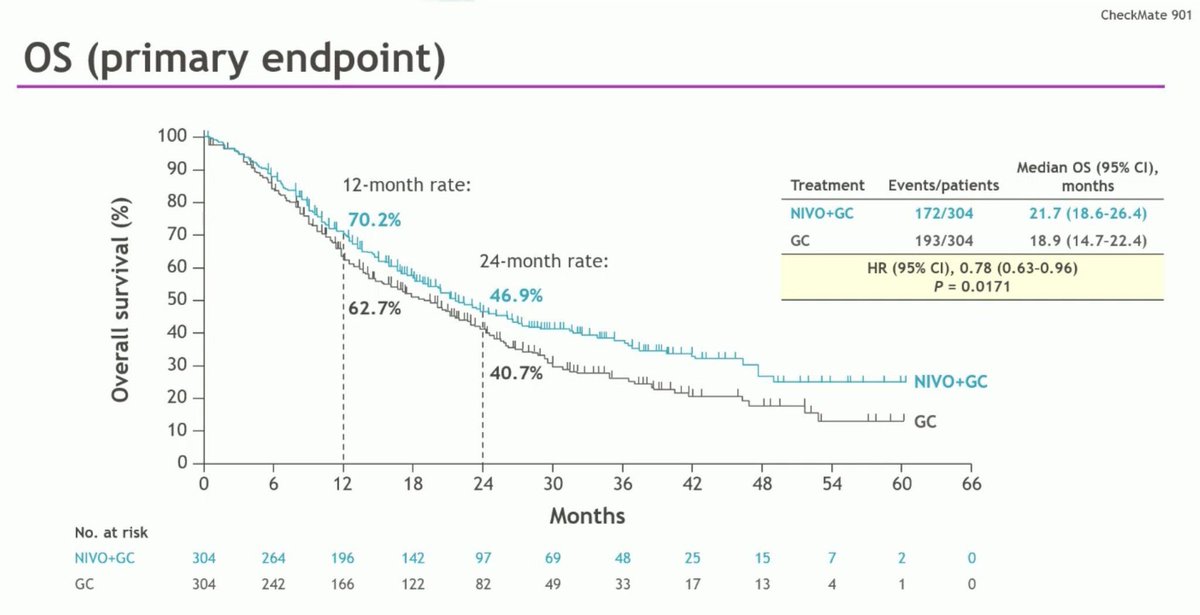
Nivo/Gem/Cis now FDA Oncology approved for 1L metastatic #bladdercancer based off #CM901 :
- Improved OS w/ Nivo (HR 0.78) 21.7mos Vs 18.9mos
- ≥ Gr3 AEs in 62% Vs 52%
- New Option (if a patient is not a candidate for EV302)
#OncTwitter #MedTwitter #gusm #OncEd


Top 5 articles of the week in CancerNetwork®
U.S. FDA FDA Oncology Professor Jacqui Shaw
ESMO - Eur. Oncology Amer. Urol. Assn.
oncodaily.com/insight/69949.…
#BreastCancer #Cancer #PancreaticCancer #ProstateCancer #ColorectalCancer #NSCLC #OncoDaily #Oncology



Thank you George from Multiple Myeloma RF, Yelak & Joe from International Myeloma Foundation and Jenny from HealthTree Foundation for strong teamwork today at Miami Myeloma MRD Meeting 2024!
Together with FDA Oncology, we continue to accelerate drug development in multiple myeloma!
#mmsm Sylvester Comprehensive Cancer Center University of Miami