Aaron Mitchell
@TheWonkologist
Medical oncologist and health services resrcher @MSKcancercenter. Oncology reimbursement reform, physician-industry COI. Views mine. @thewonkologist.bsky.social
ID:3234116877
04-05-2015 20:02:14
8,9K Tweets
3,7K Followers
763 Following


New JAMA Network Open study finds that 1 in 5 cancer drugs were approved through U.S. FDA's Real-Time Oncology Review program (shorter review, faster approval), many of which were based on surrogate markers - Did FDA ask for confirmation of clinical benefit?jamanetwork.com/journals/jaman…

Health Care Lobbying and Oncology in USA
acrobat.adobe.com/id/urn:aaid:sc… via Aaron Mitchell & Ryan Nipp, MD, MPH et al Susan Bewley James Larkin
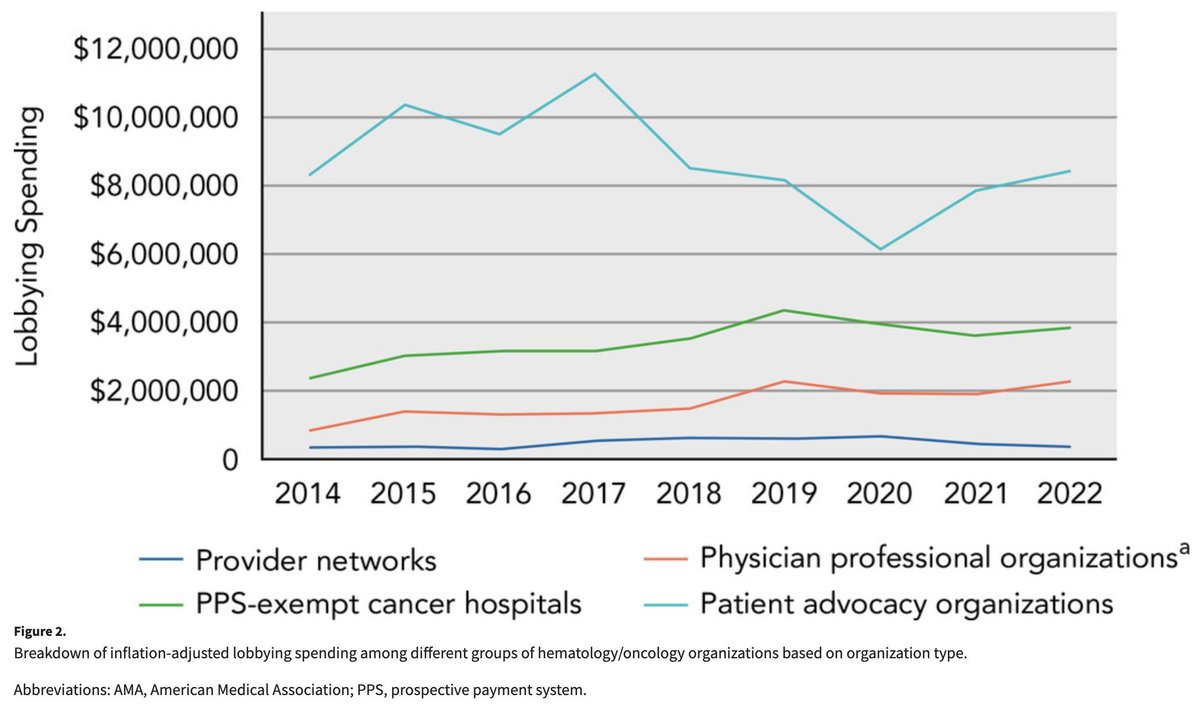

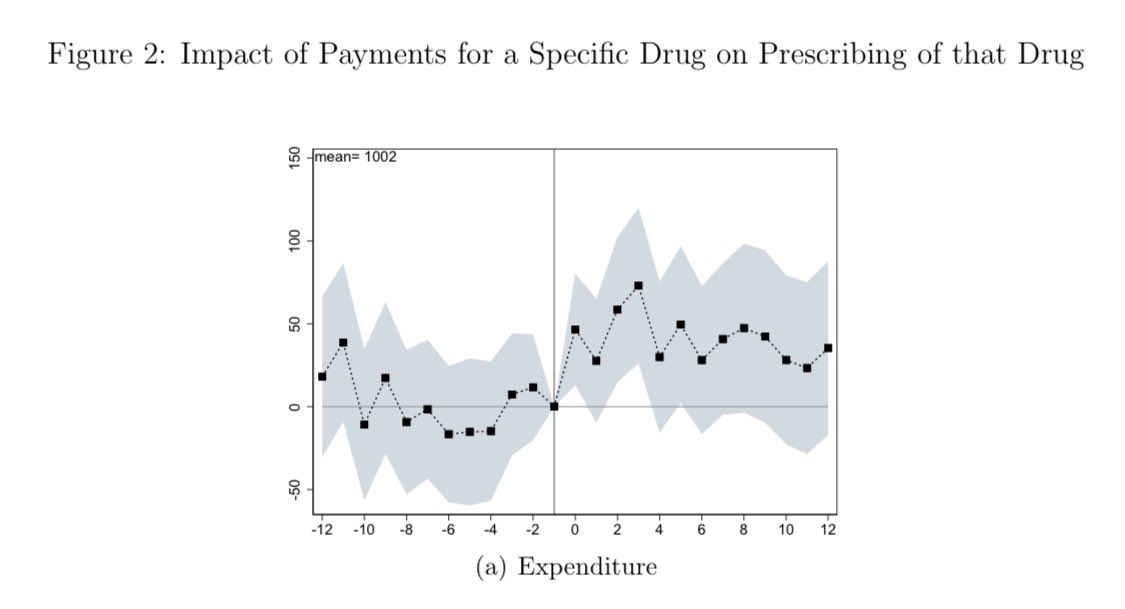
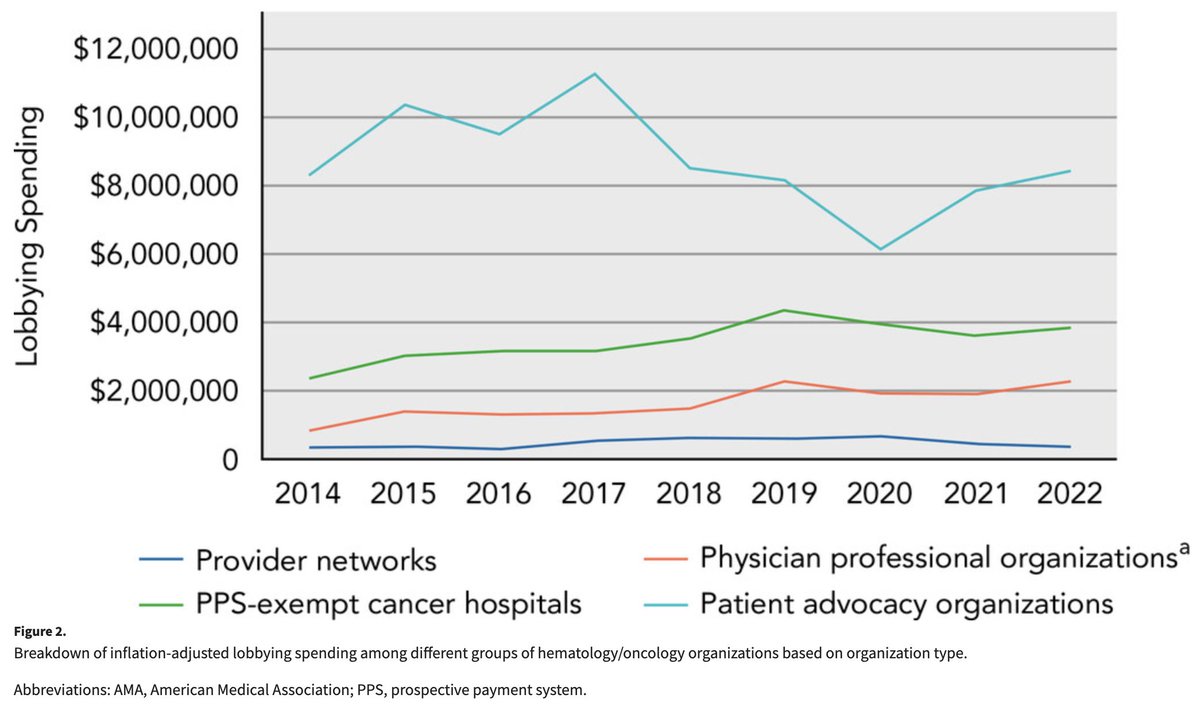
We looked at lobbying spending by oncology organizations!
Spending by advocacy organizations remains the highest, but spending by physician organizations (eg., ASCO) and PPS-exempt cancer centers has recently increased.
New in Journal of the @NCCN
Ryan Nipp, MD, MPH
jnccn.org/view/journals/…



Can we stop using BMA for SSE reduction in the mHSPC setting. Thank you Michael Morris for this important work.




People often ask me what physician-industry COI is like in Europe. My impression is that it is more similar to US than different...and they lack a system like Open Payments that at least provides some degree of transparency.
New work by James Larkin &co:
sciencedirect.com/science/articl…

💊 ❌ Majority of cancer drugs approved in the FDAs accelerated approval pathway do not improve patient survival or QOL
Important reminder for KOLs to honestly evaluate trial results & limitations
JAMA Eddie Cliff
#MedTwitter #oncology
jamanetwork.com/journals/jama/…

Check out our new study BMJMedicine to learn about how NCCN guidelines recommend oncology drug treatments with U.S. FDA accelerated approval. Yale_CRRIT Reshma Ramachandran Joseph Ross Joshua D. Wallach Josh Skydel Aaron Mitchell
bmjmedicine.bmj.com/content/3/1/e0…

Maryam Mooghali, MD, MSc and colleagues analyse the consistency of National Comprehensive Cancer Network's accelerated approval status, post approval confirmatory trials, and guideline recommendation alignment with post approval confirmatory trial results.
bit.ly/4aKFXgL



“The guidelines should explicitly state the approval pathway…of the recommended drug and provide information about the strength of evidence supporting the approval.”
Maryam Mooghali, MD, MSc in The Lancet Oncology today:
thelancet.com/journals/lanon…