
Today at Yale University at Ximena Benavides module on Ethics and Political Economy of Healthcare talking about the power of #BigTech and #BigPharma
Also had a great time yesterday presenting my work at Yale University_CRRIT discussing with law, politics, med and more scholars


Check out our new study BMJMedicine to learn about how NCCN guidelines recommend oncology drug treatments with U.S. FDA accelerated approval. Yale_CRRIT Reshma Ramachandran Joseph Ross Joshua D. Wallach Josh Skydel Aaron Mitchell
bmjmedicine.bmj.com/content/3/1/e0…

Yale_CRRIT gained another brilliant mind when hiring Melissa Barber (@mbarber.bsky.social) - if you're a staffer, this executive summary is the crème de la crème cheat sheet for evidence based talking points and policy options to address drug shortages (meaningfully).


Yale_CRRIT authors on the RTOR. Interesting evaluation of this FDA initiative. Many cancer drugs use this assessment aid.
jamanetwork.com/journals/jaman…


Hosting Reshma Ramachandran at Memorial Sloan Kettering Biostatistics Service (virtually) today, and learning a lot about the FDA accelerated approval process...and it's frequent delays.
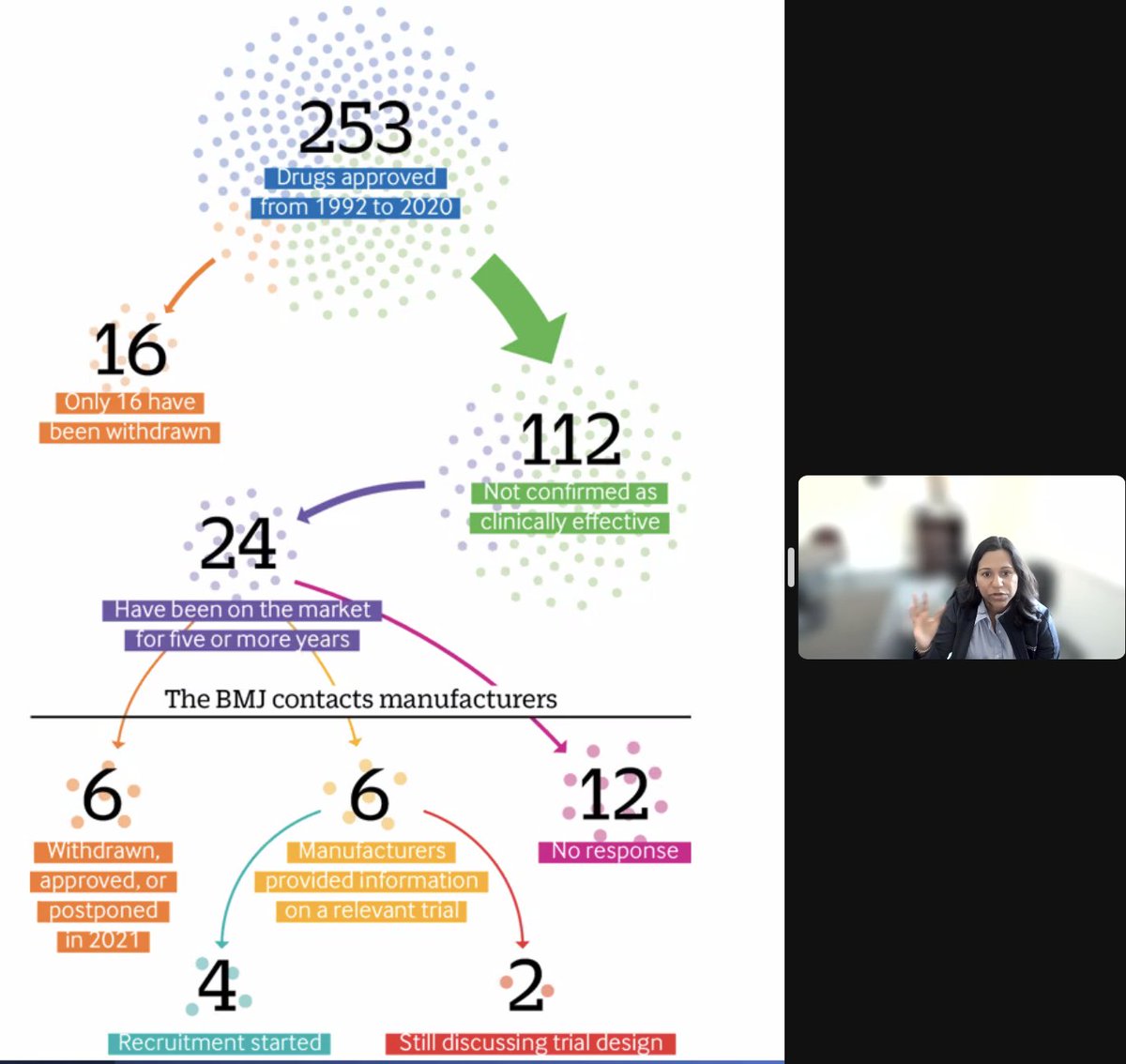
5 years on the market and no confirmation of benefit???
Yale_CRRIT

Este estudio fue elaborado por Till Bruckner, fundador de TranspariMED, en estrecha colaboración con los miembros del equipo de Cochrane, el Yale_CRRIT y el Programa de Productos Farmacéuticos y Salud de Transparency International
Resumen en #español 👏 saludyfarmacos.org/boletin-farmac…


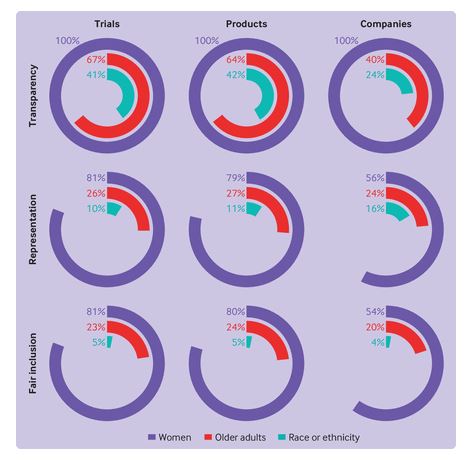
Led by Alissa Wong & Joshua D. Wallach, new Yale_CRRIT study in JAMA showed that FDA oncology approvals had similar clinical development times regardless of whether their pivotal trials had adequate representation of female, older adult, Asian, Black, or Hispanic/Latino patients.

Looking forward to speaking at Yale Yale COPPER Center Yale School of Medicine Yale School of Public Health next week! Physician-industry #coi is always a fun topic. Bonus opportunity to meet some Yale_CRRIT colleagues in person!
Thanks to Cary Gross and Dr. Dinan for the invite!


Financial conflicts of interest were common and rarely disclosed in public comments submitted to CMSGov in the National Coverage Determination process for therapeutic medical devices, Angela Lu, Robin Ji, Alex Ge, Rita Redberg, Sanket Dhruva, and colleagues at Yale_CRRIT show

Metrics, baseline scores, and a tool to improve sponsor performance on #clinicaltrial diversity ▶️ bit.ly/3wj3hjG
#MedTwitter
Tanvee Varma, MD Jennifer E. Miller Cary Gross Joseph Ross Michelle Mello Yale School of Medicine Yale_CRRIT

Really proud of this team effort! See thread below to learn more about Yale_CRRIT & Emory Public Health effort (31 systematic reviews in one paper!) to evaluate the strength of evidence for surrogate markers used to support FDA approval of drugs treating nononcologic chronic disease.

Reforming The Medical Device Recall Process—A Call For Accountability | Health Affairs buff.ly/3UMI2UH by Kushal Kadakia from @HarvardMed and Maryam Mooghali, MD, MSc Claudia See Joseph Ross Harlan Krumholz from Yale School of Medicine Yale_CRRIT CC: UCSF School of Medicine via @health_affairs

In medicine, Yale_CRRIT is exploring legal roadblocks to the public manufacturing of medicine like insulin, hoping to provide the government with the knowledge it needs to fight back against any potential challenges posed by big pharma. 7/


New JAMA Network Open study finds that 1 in 5 cancer drugs were approved through U.S. FDA's Real-Time Oncology Review program (shorter review, faster approval), many of which were based on surrogate markers - Did FDA ask for confirmation of clinical benefit?jamanetwork.com/journals/jaman…

#PublicHealth Reasonable Pricing Clauses: A First Step Toward Ensuring Taxpayers a Fair Return on their Public R&D Investment blog.petrieflom.law.harvard.edu/2023/09/28/rea… N.Chaudhry Yale_CRRIT Reshma Ramachandran @petrieflom v Zain Rizvi #PublicReturn


Join us University of Toronto Health Law, Policy & Ethics Seminar: Ximena Benavides Yale_CRRIT
Yale Law School 'Digital Clinical Trials & Private Equity Firms: Tensions Between Efficiency and Drug Evidence Access' Room P155 Jackman Law Building, 78 Queen’s Park 12.30-2 pm. law.utoronto.ca/events/health-…


Great to see new Yale_CRRIT study led by Yale School of Medicine's Guneet Janda (@reshmagar Meera Dhodapkar Joseph Ross) published!
We found that few pivotal trials supporting supplementary new indication approvals can be feasibly emulated using contemporarily available real-world data (RWD).

New JAMA study w/ Alissa, Reshma Ramachandran Jennifer E. Miller Maryam Mooghali, MD, MSc Joseph Ross!
Emory Public Health Yale_CRRIT
Are FDA-approved cancer drugs supported by pivotal trials w/ adequate representation of minoritized groups associated w/ slower clinical development times?1/7