Joseph Ross
@jsross119
Primary care physician/health policy research @YaleMed; @NCSP_Yale; healthy skeptic; @JAMA_current ed; open science: @YODAProject @Yale_CRRIT @medrxivpreprint
ID:185653786
http://medicine.yale.edu/intmed/people/joseph_ross-1.profile 01-09-2010 14:25:45
11,3K Tweets
5,9K Followers
1,4K Following


📣 Webinar recording and slides now available:
'The Yale Open Data Access Project (The YODA Project): 10 Years of Clinical Trial Data Sharing' with Joseph Ross Joseph Ross of Yale University
🔗 bit.ly/4btqF0v #pctGR


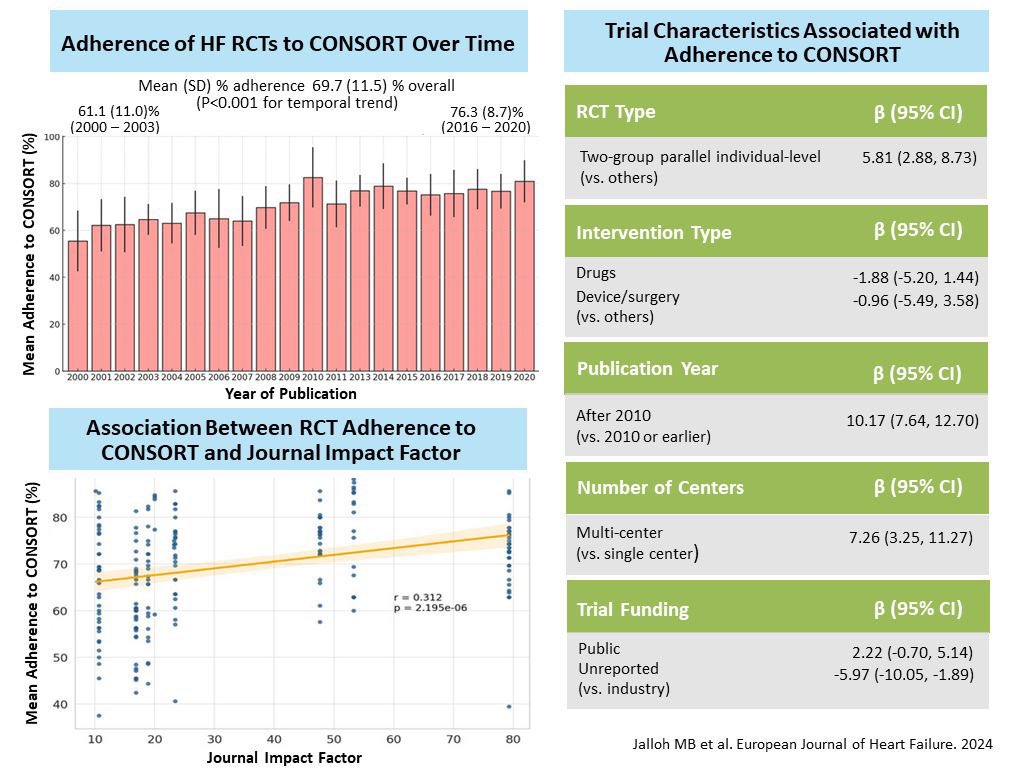
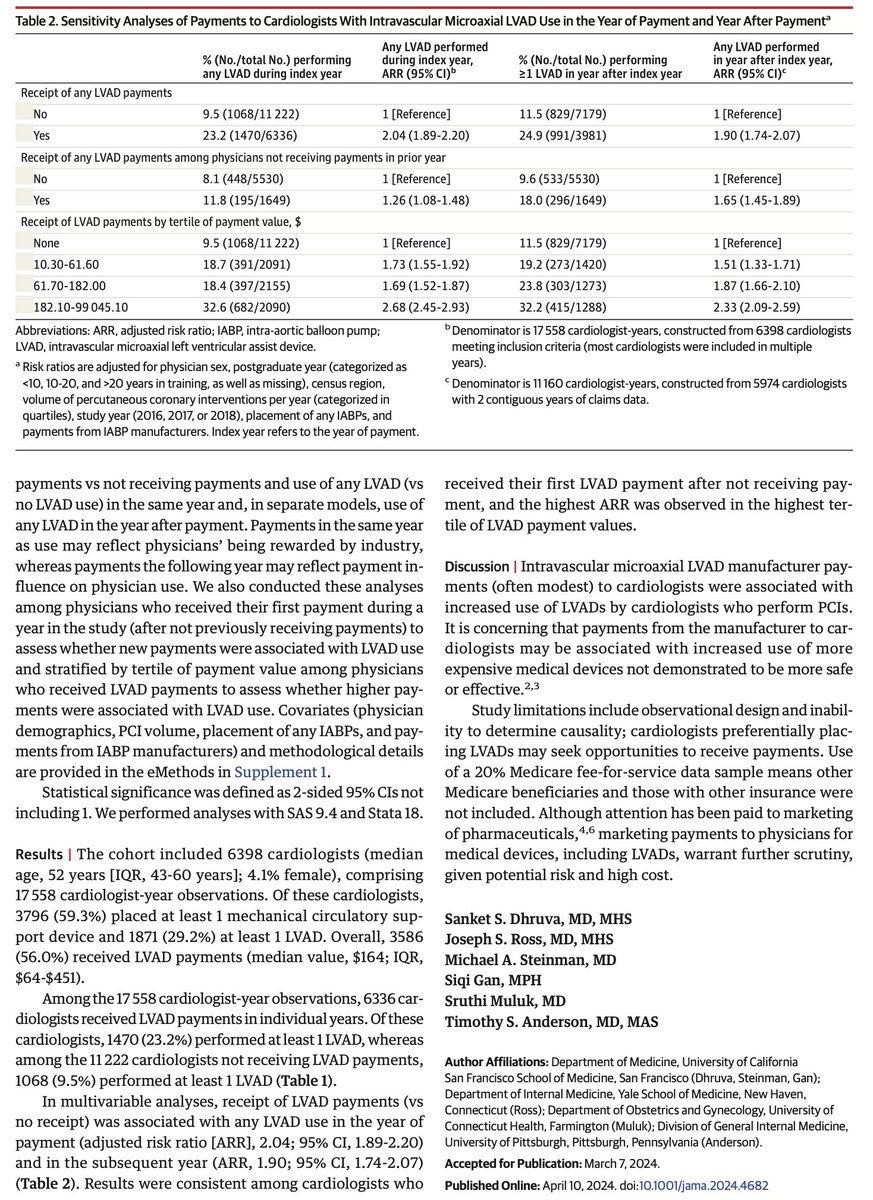
Massively important study showing once again that if you want to know the outcome of a treatment, you need to measure the outcome and not just something you believe (often wrongly) to be causally associated with it.
Raj Mehta James Lind Sam Finnikin Bishal Gyawali

Many U.S. FDA approved non-oncologic surrogate markers lack meta-analyses linking them to outcomes, says Joshua D. Wallach & Joseph Ross et al study raps.org/news-and-artic… via Jeff Craven

Study: Many surrogate markers used for pivotal trials of products to treat non-oncologic chronic diseases lack meta-analyses linking them to outcomes, raising uncertainty of their validity Joshua D. Wallach Yale_CRRIT raps.org/news-and-artic…

.Joseph Ross, Joshua D. Wallach and others have an new paper in JAMA out today seeking more FDA transparency on the use of surrogate endpoints outside of oncology - endpts.com/researchers-se…

Great thread about our paper, published today and led by Joshua D. Wallach along with several Yale School of Medicine students. Surrogate markers are increasingly being used in trials, esp those to support FDA product approval, but what’s the evidence to support their use?


In this week's PCT Grand Rounds, we welcome Joseph Ross Joseph Ross of Yale School of Medicine, codirector of the The YODA Project, to discuss the project's 10 years of experience with clinical trial data sharing. Join us! bit.ly/49CHe8L


Really sad to see that Dr Egilman passed away.
nytimes.com/2024/04/15/hea…
He hosted Cristina Candal Pedreira at Brown University Brown University School of Public Health and was a really commited and respected researcher on conflict of interest issues.
Joseph Ross



In our new study BMJMedicine, CRRIT Postdoc Maryam Mooghali, MD, MSc examined NCCN guidelines’ recommendations for oncology drug treatments with U.S. FDA accelerated approval. Reshma Ramachandran Joseph Ross Joshua D. Wallach Aaron Mitchell Josh Skydel
bmjmedicine.bmj.com/content/3/1/e0…


My thoughts on our recent JAMA paper on industry influence and Sowell's Conflict of Visions
johnmandrola.substack.com/p/our-jama-pap…
Thanks Andrew Foy Joseph Ross Ahmed Sayed and Dr. Lisa Soleymani Lehmann




This Viewpoint from Yale Law School Sahil Agrawal, MD, PhD, Joseph Ross, Reshma Ramachandran describes implications for medicine and public health if the US Supreme Court decides to overturn or narrow Chevron deference. ja.ma/4953mbE